HPR supports your R&D activities through:
-
Development and characterization of your NHP disease models
-
Preclinical evaluation of potential therapeutic candidates prior to clinical trials
-
AAALAC International standards for animal care
Our Team

CEO Message:
"Our clients are extremely satisfied with our contract studies, which were completed successfully by our technicians who are highly skilled in handling of nonhuman primates as well as rodents. In addition to our osteoarthritis, pain, and stroke models, HPR recently developed the photochemically-induced thrombosis (PIT) method to induce vascular cyclic flow reduction, a phenomenon observed in vascular thrombosis and cerebral ischemia. HPR integrated PIT with in vivo imaging technologies, including MRI and PET, to track disease progression in the same subject over time. We are fully committed to supporting your drug discovery efforts with professionalism and our extensive experiences and skill sets."
Our History
2005
Founding of Hamamatsu Pharma Research, Inc, Japan
CEO Hiroyuki Takamatsu, PhD
2013
Primate Center established
2010
Head office / Central Research Facility established
2015
Accreditation by AAALAC International
2021
Signa Explorer 1.5T (GE Healthcare) on-line at HPR, Japan
2022
CEO Takahiro Natsume, PhD
Our History
2005
Founding of Hamamatsu Pharma Research, Inc, Japan
CEO Hiroyuki Takamatsu, PhD
2013
Primate Center established
2010
Head office / Central Research Facility established
2015
Accreditation by AAALAC International
2021
Signa Explorer 1.5T (GE Healthcare) on-line at HPR, Japan
CEO Takahiro Natsume, PhD
Our History
2005
Founding of Hamamatsu Pharma Research, Inc, Japan
CEO Hiroyuki Takamatsu, PhD
2013
Primate Center established
2010
Head office / Central Research Facility established
2015
Accreditation by AAALAC International
2021
Signa Explorer 1.5T (GE Healthcare) on-line at HPR, Japan
CEO Takahiro Natsume, PhD
2022
2023
HPR has joined as a subsidiary company of CMIC HOLDINGS Co., Ltd.
Our Facilities
-
All studies are reviewed and approved by the Hamamatsu Pharma Research Animal Care and Use Committee.
-
Animal care and housing in accordance with the "Guide for Care ad Use of Laboratory Animals" (Institute for Laboratory Animal Research, 2011)
-
HPR's Primate Center, Central Research Facility, and Imaging Center are located in Hamamatsu, Japan.
Primate Center
-
8 holding rooms (up to 300 macaques)
-
4 procedure rooms
Central Research Facility
-
1 drug preparation room
-
1 cell culture room
Imaging Center
-
MRI room



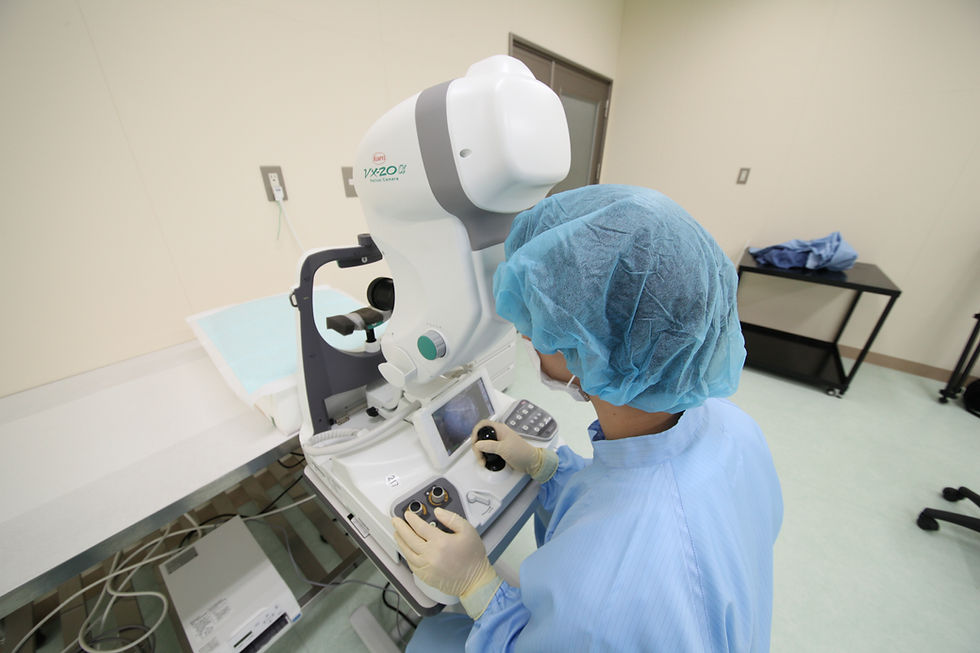
Ophthalmology examination


Blood chemistry analysis


Pharmacological study room

HPR publication
